Depo-Provera Lawsuits Surge: Birth Control Injection Linked to Brain Tumor Risk
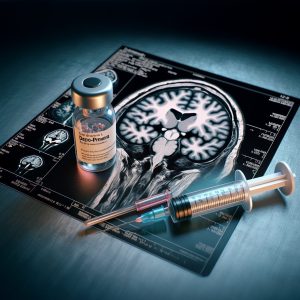
Depo-Provera is one of the most widely prescribed contraceptives on the market.
Across the country, millions of women rely on Depo-Provera. According to some estimates, more than a quarter of all sexually active women in the United States have taken this drug at least once. When it comes to preventing unwanted pregnancies, Depo-Provera is highly effective, and that is not up for debate. And, since it only needs to be taken once every three months, it also represents a convenient alternative to oral birth control.
However, even though Depo-Provera’s reputation as a contraceptive is rock-solid, it is not risk-free.
Ever since it was first introduced a half-century ago, women have reported a wide range of side effects. Most of these complications are relatively minor, with the most common including complaints of stomach aches, recurring headaches, and fatigue. Scientists have since uncovered evidence that strongly suggests a causal relationship between Depo-Provera and meningioma, a type of tumor that can form in the membranous layer surrounding the brain.
Here is what you need to know about:
Depo-Provera
Depo-Provera is a type of injectable birth control that is administered once every three months.
The active ingredient in Depo-Provera is medroxyprogesterone acetate, a synthetic hormone. It helps prevent pregnancy by suppressing ovulation and triggering cervical mucus production. Taken as prescribed, Depo-Provera can effectively shut down ovulation, acting as an almost-total safeguard against pregnancy.
Birth Control and Brain Tumors
A recent study published in the British Medical Journal found that the “prolonged use of medrogestone, medroxyprogesterone acetate [the active ingredient in Depo-Provera], and promegestone was found to be associated with an increased risk of meningioma.”
Meningiomas are tumors that can form in the meninges, a series of three membranes encasing the brain and spinal cord. Although meningiomas are tumors, they are rarely cancerous; only between 1% and 10% of all diagnoses are malignant. However, while most meningiomas are not cancerous, they can still be very dangerous. Left untreated, these tumors can grow and expand, potentially putting pressure on nearby cerebral tissue, blood vessels, and nerve clusters. This can lead to severe and debilitating neurological problems.
Notably, most meningiomas form slowly. It can take years for symptoms to become noticeable and problematic enough to see the doctor. Even if you do start feeling sick, the initial signs may be too vague for diagnosis, with some of the most common including headaches, dizziness, and hearing loss.
Your Rights After A Meningioma Diagnosis
If a pharmaceutical company has reason to believe that a medication could cause serious side effects, they are legally obliged to warn patients of the risk. However, while medical researchers have had concerns about Depo-Provera and meningioma for decades, Pfizer did not update its warning labels until December 2025. As a result, most women who take or have taken Depo-Provera were never warned about the risk of brain tumors.
You could be entitled to compensation if you meet the following criteria:
- You were prescribed Depo-Provera;
- You took Depo-Provera for an extended period of time; and
- You have been diagnosed with meningioma or another serious condition caused by your medication.
If you think you could have a case, act fast: if you delay, critical evidence could go missing, or the statute of limitations applicable to your claim could lapse. Contact the Dietrich Law Firm P.C. today at 1-866-529-5334 to speak to a Depo-Provera injury lawyer in New York and schedule your 100% free, no-obligation consultation as soon as possible.
 Buffalo Personal Injury Lawyer News
Buffalo Personal Injury Lawyer News